Corrosion in metals deteriorates the metal parts due to chemical reactions between metal and the surrounding environment. In this article, we will discuss what is galvanic corrosion, its applications, and how to prevent it using a galvanic corrosion chart? We suggest you also read this article on anodizing.
What is Galvanic Corrosion?
Galvanic/Dissimilar Metal or Bimetallic Corrosion is a type of electrochemical corrosion, where a material corrodes if it comes in contact with another material in the presence of an electrolyte.
During product design, engineers should select material ensuring the dissimilar metal corrosion has the minimum or a positive impact on product function. They should always refer galvanic corrosion chart during the material selection process.
How does Galvanic Corrosion Occurs ?
Galvanic corrosion between different materials occurs due to galvanic reactions between two non-similar electrochemical potential materials.
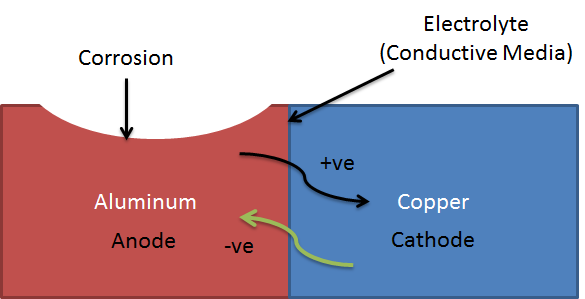
When two different metals with different electrochemical potential come in contact with each other. The potential difference between materials causes the flow of electrons from the cathode to the anode.
This flow of electrons causes the deterioration of the material. The higher the flow of electrons from the cathode to anode, the higher will be the corrosion.
Galvanic Corrosion Chart
| S.No. | Material | Anodic Index (V) |
|---|---|---|
| 1 | Gold, Gold-platinum alloy | 0 |
| 2 | Rhodium plated on silver-plated copper | 0.05 |
| 3 | Silver, monel metal, High nickel-copper alloys | 0.15 |
| 4 | Nickel, titanium alloys, monel | 0.3 |
| 5 | Copper, low brasses or bronzes, silver solder, German silvery high copper-nickel alloys, nickel-chromium alloys | 0.35 |
| 6 | Brass and bronzes | 0.4 |
| 7 | High brasses and bronzes | 0.45 |
| 8 | 18% chromium type corrosion-resistant steels | 0.5 |
| 9 | Chromium plated, tin plated, 12% chromium type corrosion-resistant steels | 0.6 |
| 10 | Tin-plate, tin-lead solder | 0.65 |
| 11 | Lead, high lead alloys | 0.7 |
| 12 | Aluminium, wrought alloys 2000 Series | 0.75 |
| 13 | Iron, wrought, gray or malleable, plain carbon and low alloys steels | 0.85 |
| 14 | Aluminium (other than 2000 Series), cast alloys of silicon type | 0.9 |
| 15 | Aluminium, cast alloys other than silicon type, cadmium, plated and chromate | 0.95 |
| 16 | Hot-dip-zinc plate; galvanized steel | 1.2 |
| 17 | Zinc, wrought; zinc-base die-casting alloys; zinc plated | 1.25 |
| 18 | Magnesium & magnesium-base alloys, cast or wrought | 1.75 |
| 19 | Beryllium | 1.85 |
Note : Values given here are for indicative purpose only. Final decision on material and finish selection shall be done based on tests.
Example of Galvanic Corrosion Between Aluminum and Steel
Galvanic corrosion occurs when two dissimilar metals are in contact electrically in the presence of an electrolyte. During this process, corrosion occurs on the anode, whereas the cathode is protected.
For example when steel (anodic index = 0.6) (refer galvanic corrosion chart) comes in contact with aluminum (anodic index = 0.75) (refer galvanic corrosion chart). Aluminum works as an anode and steel as a cathode. Therefore corrosion occurs on aluminum because it has a higher anodic index value and works as an anode.
How to prevent Galvanic Corrosion?
We can prevent bimetallic or galvanic corrosion if we can avoid any of the following three dis-similar metal corrosion conditions.
- Electro-chemically dissimilar metals with different anodic indexes.
- Dissimilar metals must be in electrical contact.
- Metals must be exposed to the electrolyte for the movement of metal ions from anodic to cathodic metal.
But in the real scenario, it is very difficult to avoid these conditions in product design. Therefore following points are considered during product design to reduce the impact of galvanic corrosion.
1) Incorporate Insulation in between dissimilar Metals
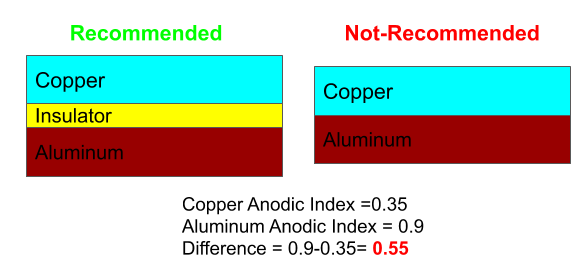
Insulation between dissimilar metals helps in preventing bimetallic corrosion because electrical contact between dissimilar metals is a prior condition for galvanic corrosion.
2) Absence of Electrolyte

Dissimilar metal corrosion can be prevented by ensuring electrolyte/water or any other conductive media is not in contact with dissimilar materials. Because in this way movement of metal ions from anodic to cathodic metal can be prevented.
3) Selection of materials closer in galvanic series

We should select the material during part design by referring galvanic corrosion chart to prevent galvanic corrosion. The galvanic corrosion chart indicates anodic index values for different materials.
Engineers always recommend the minimum possible difference of anodic index for different materials because the higher the differences in anodic index values, the higher the corrosion rate.
4) Size of Cathode and Anode in dissimilar Metals
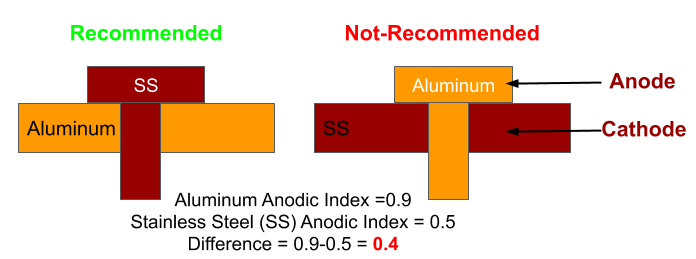
Smaller the surface area of the anode compared to the cathode, Higher will be the concentration of flow of electrons from anode to cathode. As a result, the rate of corrosion will be high.
Therefore dissimilar material corrosion can be prevented by avoiding small anodes in contact with larger cathodes.
5) Metallic and non metallic coating
Non-Sacrificial coatings such as powder-coating or paint, work as a barrier and reduce galvanic corrosion. But these coatings are not as effective as sacrificial coatings in preventing dissimilar metal corrosion.
Advantages of Galvanic Corrosion?
Galvanic reactions between two different metals can have the following advantages while used for engineering applications.
- Anodic metallic coatings provide sacrificial galvanic corrosion protection to the base material. Metallic coating sacrifices itself to protect the base material. For example, Zinc coating on steel prevents galvanic corrosion of steel.
- Even damaged sacrificial coating continues to protect the base material. The extent of corrosion protection depends on coating material and thickness. For example, 12-micron zinc plating will provide more corrosion resistance compared to 8-micron coating.
Frequently Asked Questions: FAQ
Following three prior conditions are required for galvanic corrosion to occur.
- Electro-chemically dissimilar metals with different anodic indexes.
- Dissimilar metals must be in electrical contact.
- Metals must be exposed to the electrolyte for the movement of metal ions from anodic to cathodic metal.
If any of the above three conditions can be avoided in product design. Galvanic corrosion can be prevented.
Zinc will corrode first because zinc has a higher anodic index compared to iron.
Yes, the size of a part has a considerable impact on the extent of galvanic corrosion.
The smaller the surface area of the anode compared to the cathode, the higher will be the concentration of flow of electrons from anode to cathode. As a result, the rate of corrosion will be high.
Therefore dissimilar material corrosion can be prevented by avoiding small anodes in contact with larger cathodes.
To sum up, Galvanic Corrosion is a type of electrochemical corrosion. Where a material corrodes if it comes in contact with another material in the presence of an electrolyte. This phenomenon of metals is used to prevent corrosion of base metal by providing sacrificial coatings.
We will keep adding more information on dissimilar material corrosion. Please add your comments or questions on galvanic corrosion chart in the comment box.
I get all this but still cannot figure out what material I should choose for the plus-nuts I need to install inside my Ford Transit Connect van on the body (made of “boron steel”, which is I think carbon low-alloy steel, with 0.85V anodic index) to prevent galvanic corrosion. Options for plus-nuts are: stainless steel (0.5V), zinc-coated carbon steel (1.25V on 0.85V), and aluminum alloy, typically 6061 which is a silicon type (so index is 0.9V). Some level of humidity will be present just from a person living in it. I will try to use the existing (OEM painted) holes in the body so as to not generate uncoated debris falling in between the 2 sheets of the body and corroding. Then I also need to choose the bolts that will thread into those plusnuts to mount wooden wall planking and furniture. Automotive screws and bolts are stainless steel (0.5V) or zinc-coated (1.25V). My best guess is: Aluminum-Silicon-alloy plusnuts (0.9V) with the same material bolts/screws. But van conversion people are using zinc-plated steel plusnuts and whatever bolts–majority of people I asked don’t even know about galvanic corrosion or say its negligible in an indoor situation). A tech answer would be much appreciated!
Use the same material. So, for Boron Steel use zinc-coated fasteners. There is a reason they are the most commonly used fasteners in the automotive industry. Do not use aluminum fasteners in steel application, first for strength reasons and second no one does that.
Helpful Blog! We appeared to be just discussing there are lots screwy outcomes at the issue you now purely changed my very own belief. Thank you really significantly a perfect write-up